Introduction
For those of you who follow my work, you’re likely well acquainted with the fact that I am highly critical of climate models, extreme weather event attribution, and climate change alarmism. Even so, as an aspiring meteorologist who deals with the atmosphere on a daily basis, I am cautious about which parts of global warming theory that I reject.
While most scientifically credentialed climate change skeptics, and/or people who have studied the issue independently accept that the atmosphere has some sort of “greenhouse effect,” which ultimately keeps the lower atmosphere warm enough to inhabit and upper atmosphere cold, there never seems to be a shortage of people who claim that there is no such thing as the “greenhouse effect.” Perhaps I accept the idea because I deal with the greenhouse effect whenever I make a temperature forecast (i.e. water vapor and clouds), and others don’t because they have no clue how to forecast the weather… just a thought (?).
Those who claim that there is “no such thing” as the greenhouse effect will often claim one, two, or any combination of these four things:
- Adiabatic compression of the air (atmospheric pressure) is what causes the atmosphere’s temperature profile.
- Greenhouse gases emit infrared radiation just as fast as they absorb it.
- Greenhouse theory violates the 2nd Law of Thermodynamics, which essentially states that the net flow of thermal energy is from an object of higher temperature to an object of lower temperature.
- There is no way on Earth (pun intended) that greenhouse gases, which constitute a small part of the atmosphere, can cause the “greenhouse effect.”
The theory that adiabatic compression causes the thermal structure of the atmosphere is a quite compelling argument, but there are a number of flaws with this theory, as I’ll address in this article. Additionally, while the 2nd Law of Thermodynamics and greenhouse gas concentration arguments do seem valid, I’m going to explain why the greenhouse effect does not violate the Laws of Thermodynamics and why the kinetic theory of gases explains why such small concentrations of these gases results in warming.
Before I embark on this journey however, I would like to set the record straight that the atmospheric “greenhouse effect” does not actually work like a closed-roof greenhouse that you grow plants in, which suppresses convection. One of my best friends growing up has a greenhouse and we would check up on the plants on a daily basis during the summer; I’m well aware of the differences between it and the open atmosphere. I am sure some commenters will complain about the misnomer, but there’s nothing I can do about it. The terminology has stuck over the years, so it’s time to suck it up, let it go, and think outside the box.
Greenhouse Gases
Anything that has a temperature will give off electromagnetic radiation, or light.¹ This radiation can either be in shortwave form, which is visible light, or in longwave form, which is infrared, depending upon the intensity and amount of energy.¹ The sun gives off shortwave radiation, which is of higher energy, and the Earth emits longwave energy, which is cooler and of lower energy.¹
A greenhouse gas is a gas that is able to absorb and emit outgoing longwave infrared radiation, but is transparent to incoming shortwave energy from the sun. The collection of these gases in the atmosphere is responsible for the “greenhouse effect,” as I’ll discuss later. The prominent greenhouse gases in the Earth’s atmosphere include water vapor and carbon dioxide, though methane, nitrous oxide,² and ozone are also greenhouse gases.
Gas molecules that are chemically bonded by more than two atoms are loosely held together and are generally good absorbers [and therefore good emitters] of infrared radiation.² ³ They have complex vibration modes which interact with the radiation.² ³ It just so happens that gases like water vapor and carbon dioxide are composed of three atoms each.³ Nitrogen, oxygen, and argon are not greenhouse gases because molecules that are diatomic (containing two atoms of the same element) and monatomic (one atom) do not have vigorous vibration modes.² ³ They only vibrate by moving closer together and then further apart.³ Thus, they’re unaffected by infrared radiation.
The “Greenhouse Effect”
The ultimate source of energy that is supplied to the Earth and its atmosphere comes from the sun.⁴ In order for Earth to maintain some reasonable state of temperature equilibrium, all of the sun’s shortwave radiation must be sent back into outer space.⁴ Earth emits this energy in longwave form because it’s not as hot as the sun.¹
Remember, the temperature of anything is solely a function of how much energy is being gained versus how much energy is being lost.⁵ ⁶ Thus, in order for the temperature on Earth to remain constant (which it almost never has or ever will contrary to what Hockey Stick proponents and climate activists want you to believe), energy in must equal energy out.⁴ ⁵ If the energy gain exceeds the losses, temperature goes up and if energy loss exceeds the gain, the temperature decreases.⁵
While there are a wide array of gases mixed like a salad in the atmosphere, as shown in Table 1 below, the gases that are responsible for the atmosphere’s temperature profile are “greenhouse gases.” Because of hydrostatic balance (a function of gravity and the upward-directed pressure-gradient force), air density and therefore the number of air molecules decrease with height exponentially.
| Gas (Symbol) | Content (%) in Atmosphere |
| Nitrogen (N₂) | 78.084% |
| Oxygen (O₂) | 20.946% |
| Argon (Ar) | 0.9340% |
| Carbon Dioxide (CO₂) | 0.041332% (As of April 2019) |
| Neon (Ne) | 0.001818% |
| Helium (He) | 0.000524% |
| Methane (CH₄) | 0.000187% |
| Krypton (Kr) | 0.000114% |
| Water Vapor (H₂O) (wet composition) | 0.00 to 3.00% (depends on weather and region) |
While greenhouse gases comprise a remarkably small fraction of the atmosphere’s chemical makeup as shown in Table 1 above, their speed and motion, which increases as a response to absorbing longwave radiation, and subsequent transfer of heat from one greenhouse gas molecule to another, results in continuous collisions leading to more emitted infrared radiation.⁷ This is part of the kinetic theory of gases.
A good bit of the radiation that is injected into the atmosphere by solar heating is reflected back into outer space, but things like greenhouse gases and clouds inhibit most of that radiation from escaping as fast as it normally would. Trenberth et al (2008) demonstrated this, and they have a very good diagram that goes along with it in Figure 1 below.⁸
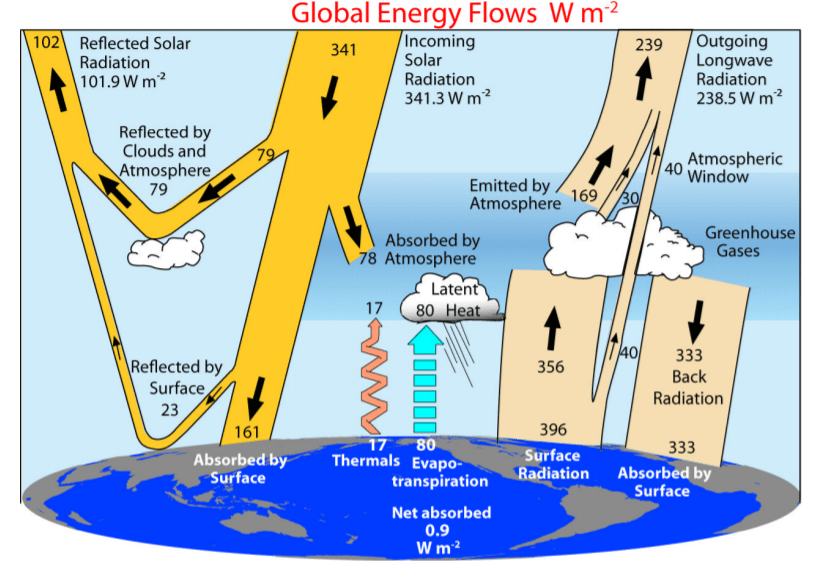
Anything that absorbs infrared radiation is also good at emitting it.⁹ While the rates of energy gain are not very dependent on temperature, the amount of energy loss is.¹⁰ The warmer an object or something is, the more infrared radiation it emits to try and maintain a constant temperature.
Since greenhouse gases are good absorbers and emitters of infrared radiation photons, any radiation that is emitted will occur in all directions: upwards into outer space, sideways, and downwards.⁹ The radiation that is emitted upward into outer space cools the mid and upper levels of the atmosphere because there are fewer molecules, while the downgoing (a.k.a. back radiation) warms the lower layers because that’s where most of the atmosphere’s mass is.⁹
The reason that the lower layers are warmed is because all in all, those greenhouse gases slow the rate of cooling by limiting how much infrared radiation is lost to outer space.¹¹ One way of increasing the temperature of something is by slowing the rate of energy loss, which is what the atmospheric greenhouse effect does. Temperature doesn’t necessarily need to be increased by increasing energy gain.
It has been calculated that without greenhouse gases in the atmosphere, Earth’s average surface temperature would be about 0°F (-17.8°C), but this number is just theoretical.⁹ If there were no greenhouse gases, mainly water vapor, Earth’s average surface temperature would probably plummet to those levels, yes. However, those calculations don’t consider the fact that a colder planet with water, especially one that cold, would have more ice at its poles, more glaciers, more ice sheets, etc… increasing the surface albedo leading to a positive feedback loop.⁹
Those who suggest that there is no greenhouse effect will often claim that the theory violates the 2nd Law of Thermodynamics, which essentially states that the net flow of energy is always from warmer to colder objects. They claim that because the upper atmosphere is colder, it can’t emit back radiation to the lower levels and cause it to be warmer. This isn’t true. It doesn’t matter what the temperature difference is between two objects, there is always an exchange of energy, however, the net flow is always from warmer to colder temperatures.¹¹ This is the case in the atmosphere as well. The net flow of energy is from warmer to colder temperatures, which is why the temperature decreases with height in the atmosphere. Any planet that has greenhouse gases in its atmosphere will have a lapse rate (temperature change with height).⁹ This lapse rate, which is caused by the greenhouse effect, is responsible for atmospheric convection and ultimately our weather.⁹ Without the greenhouse effect, there would be no convective overturning and therefore no weather.⁹
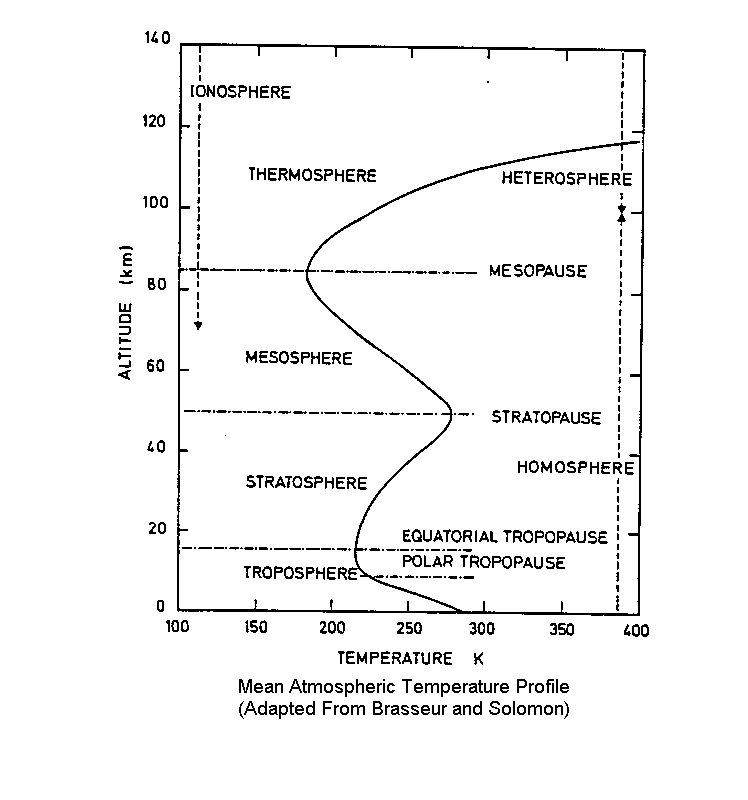
Other people suggest that adiabatic compression as a result of atmospheric pressure is responsible for the temperature structure of the atmosphere. The issue with this, however, is that adiabatic theory only explains temperature change with altitude, it does not tell us anything about what the actual temperatures will be. You can not compute temperature just by looking at the temperature change, which makes the “non-greenhouse” theory incorrect. Additionally, the atmospheric pressure profile of the atmosphere (which decreases exponentially with height) does not explain why the temperatures in the stratosphere increase with altitude as shown in the figure adapted from Brasseur and Solomon (2005).
Before I make my closing remarks, I’d like to add one more tidbit of information that may spark some discussion. If there is no greenhouse effect, as some skeptics claim, why is it that cloudy nights are warmer than clear ones? Remember, clouds are the biggest greenhouse forcings aside from water vapor.
If you buy into the pressure theory, I challenge you to have a solid, alternative explanation as to why cloudy nights are warmer than clear nights. Obviously, air pressure does not explain this occurrence, because cloudy nights are generally associated with lower atmospheric pressure, not higher atmospheric pressure.
Closing Remarks
Nearly 11 years ago, Dr. Richard Lindzen, a prominent atmospheric scientist, warned skeptics in his 2nd annual Heartland conference that climate change skeptics need to be very careful about which parts of global warming theory that they dispute, question, or deny. The underlying physics are sound and easily explain the temperature stratification of the atmosphere.
While the so-called “greenhouse effect” does not work like a real greenhouse, the terminology has remained, so just deal with it. The processes discussed above do not violate the laws of thermodynamics, and atmospheric pressure does not explain why temperatures increase in the stratosphere.
If you ask almost any climate change skeptic or lukewarmer like myself, especially those who are trained scientists, they will be quick to tell you that the climate change dialogue and debate is about climate feedbacks, extreme weather events, and solutions to the problem, if there even is one (which I don’t think there is). There is little dispute within the scientific community about the “greenhouse effect,” it’s well-established physics that has been published over and over again and proven in various laboratory studies.
Hopefully, this will spark some conversation in the comments section, which will remain open as long as it doesn’t result in foul language or name calling.
REFERENCES
[1] “Longwave and Shortwave Radiation.” North Carolina Climate Office. Accessed February 15, 2020. https://climate.ncsu.edu/edu/RadiationTypes.
[2] “The Greenhouse Effect.” UCAR Center for Science Education, 2011. Accessed February 15, 2020. https://scied.ucar.edu/longcontent/greenhouse-effect.
[3] “Molecules Vibrate.” UCAR Center for Science Education, 2012. Accessed February 15, 2020. https://scied.ucar.edu/molecular-vibration-modes.
[4] “Earth’s Energy Balance.” North Carolina Climate Office. North Carolina Climate Office. Accessed February 15, 2020. https://climate.ncsu.edu/edu/EnergyBalance.
[5] Ackerman, Steve. “The Earth’s Radiation Energy Balance.” WXWISE ERBE. University of Wisconsin–Madison. Accessed February 15, 2020. http://cimss.ssec.wisc.edu/wxwise/homerbe.html.
[6] Spencer, Roy W. “What Determines Temperature?” Roy Spencer, PhD, April 9, 2013. Accessed February 15, 2020. http://www.drroyspencer.com/2013/04/what-determines-temperature/.
[7] Spencer, Roy W. “FAQ #271: If Greenhouse Gases are such a Small Part of the Atmosphere, How Do They Change Its Temperature?” Roy Spencer, PhD, June 17, 2010. Accessed February 15, 2020. http://www.drroyspencer.com/2010/06/faq-271-if-greenhouse-gases-are-such-a-small-part-of-the-atmosphere-how-do-they-change-its-temperature/.
[8] Trenberth, Kevin E., John T. Fasullo, and Jeffrey Kiehl. “Earths Global Energy Budget.” Bulletin of the American Meteorological Society 90, no. 3 (2009): 311–24. https://doi.org/10.1175/2008bams2634.1. (PDF here)
[9] Spencer, Roy W. “What Causes the Greenhouse Effect?” Roy Spencer, PhD, June 13, 2015. Accessed February 15, 2020. http://www.drroyspencer.com/2015/06/what-causes-the-greenhouse-effect/.
[10] Spencer, Roy W. “Misunderstood Basic Concepts and the Greenhouse Effect.” Roy Spencer, PhD, January 1, 2013. Accessed February 15, 2020. http://www.drroyspencer.com/2013/01/misunderstood-basic-concepts-and-the-greenhouse-effect/.
[11] Spencer, Roy, W. “What is the greenhouse effect?” WeatherQuestions.com, December 14, 2019. Accessed February 15, 2020. https://weatherstreet.com/weatherquestions/What_is_the_greenhouse_effect.htm.
Categories: Climate, Greenhouse Effect
There is a greenhouse effect but it is a minor player. There is an alternate hypothesis of what keeps Earth warm:
https://wryheat.wordpress.com/2014/12/07/what-keeps-earth-warm-the-greenhouse-effect-or-something-else/
See also:
https://wryheat.wordpress.com/2018/12/17/the-broken-greenhouse-why-co2-is-a-minor-player-in-global-climate/
LikeLike
Click to access Robinson2014_0.1bar_Tropopause.pdf
LikeLike
There is a well-measured 65mW/m^2 geothermal flux over continental crust. Plug that value into the Stefan-Boltzmann equation T=64.8×Q^0.25, where Q is the flux. Compare the result with the temperature deficit between empirical measurements and those calculated with Stefan-Boltzmann and the solar flux. Then read my book. And let’s have a cup of coffee.
LikeLike
1. The Trenberth et al. paper was 2009, not 2008. No need to thank me. All in a day’s work.
2. Dr. Tinker, compare the 87 W/m^2 mean global geothermal flux with the 240 W/m^2 sunlight absorbed by the climate system.
The Stefan-Boltzmann equation is properly F = sigma T^4, where F is the flux density emitted by an object, sigma the Stefan-Boltzmann constant (5.670373 x 10^-8 W/m^2/K^4), and T the absolute temperature. If you invert it for temperature, you get T = (F / sigma)^0.25, or if you want to take out the constant, you indeed get about 64.8, but Q has to be in watts per square meter, not milliwatts. Your Q is 0.065 W/m^2, which would bring Earth to a temperature of 32.7 K.
It would NOT cause the 33 K difference between Earth’s 255 K radiative equilibrium temperature and its 288 K surface temperature, because you’ve got the wrong value for Q (or F). You should be comparing Q = 240.065 with 240.000, and that gives you a temperature difference of 255.069 – 255.051 = 0.018 K, not 33 K. You set up the problem incorrectly.
LikeLike